A new map for manufacturing
In Pharmaceutical and medical device manufacturing, the United States and parts of Europe remain centers for high-value production, while China and India continue rapid scale-up. At the same time, smaller hubs — from Ireland to Singapore — are attracting disproportionate investment for specialized capacity, favorable tax regimes, skilled labor and fast turnaround. Yet despite advances in automation, clean-room engineering and cold-chain logistics, executives say the biggest bottlenecks in new builds and cross-border production are often administrative: permits, approvals and paperwork.
Top countries for pharma manufacturing
United States
The United States continues to anchor advanced pharmaceutical manufacturing, particularly for complex products where talent, quality systems, and regulatory maturity matter.
Source: Grand View Research (U.S. biologics manufacturing outlook)
Germany
Germany is frequently cited as a leading pharmaceutical producer and exporter, supported by a deep industrial base and strong manufacturing infrastructure.
Source: Germany Trade and Invest (Pharmaceutical industry in Germany)
Switzerland
Switzerland’s Basel region remains one of the world’s best-known life sciences clusters, supporting high-value manufacturing and a dense ecosystem of suppliers and expertise.
Source: Canton of Basel-Stadt (Industry and companies overview)
Ireland
Ireland has developed into a major export-oriented pharmaceutical manufacturing center, with a strong multinational presence and a mature life sciences workforce.
Source: IDA Ireland (Pharmaceutical manufacturing in Ireland)
India
India continues to expand its role as a global pharmaceutical manufacturing hub, especially in large-scale production and cost-competitive supply.
Source: Industry overview (MedXDRG global pharma location summary)
Singapore
Singapore has positioned itself as a global biopharma hub, emphasizing advanced manufacturing capacity, investment readiness, and specialized talent.
Source: Singapore Economic Development Board (Biotech and pharmaceuticals)
Top countries for medical device manufacturing
United States
The United States remains a leading center for medical device manufacturing, spanning both high-end innovation and scaled production.
Source: Electronic Manufacturing Service (Where most medical devices are manufactured)
Germany
Germany’s reputation for precision engineering and dense medtech clustering continues to make it a key location for device production.
Source: Alpha Sophia (Leading medical device companies by country)
China
China remains a major device manufacturing center, particularly for high-volume production categories supported by broad supplier density.
Source: Electronic Manufacturing Service (Where most medical devices are manufactured)
Mexico
Mexico’s device manufacturing footprint has grown alongside nearshoring, with clusters that support North American supply chains and quick turnaround.
Source: Tijuana EDC (Medical device cluster overview)
Costa Rica
Costa Rica has built an export-oriented medical device industry recognized as a significant global producer and exporter.
Source: U.S. International Trade Commission (Costa Rica medical devices executive briefing)
Japan
Japan remains a major medical device market and manufacturing base, supported by a large and organized industry presence.
Source: International Trade Administration (Japan medical devices country commercial guide)
South Korea
South Korea continues to invest in and expand its medical device industry, supported by coordinated industry development efforts.
Source: Medical Korea / KHIDI (Industry development overview)
The overlooked constraint: documents that must travel before products can
When companies establish overseas operations, register products, authorize distributors, or move regulated goods internationally, they often face country-specific requirements for document acceptance. Corporate papers, powers of attorney, quality certificates, and trade documents can become gating items.
A common point of friction is timing. Quality and regulatory teams may be ready, facilities may be validated, and commercial plans may be set, but a missing authentication step can delay the onboarding process with a local partner.
“Manufacturing decisions are global, but compliance is local,” one operations director said. “And local rules tend to be unforgiving.”
How certificate authentication fits into global production
For U.S.-manufactured or marketed products, expanding into global markets requires authenticated documentation to support product registration in each target country. Local operating companies must submit these documents — bearing either an apostille (for Hague Convention countries) or full embassy legalization (for non-Hague countries) — to their respective health authorities as part of the registration process. These submissions are often time-bound, making the timely delivery of authenticated documents critical to ensuring business continuity. If these documents are not submitted on schedule, global product launches can be delayed, shipments can be held at customs, and the entire supply chain can be disrupted.
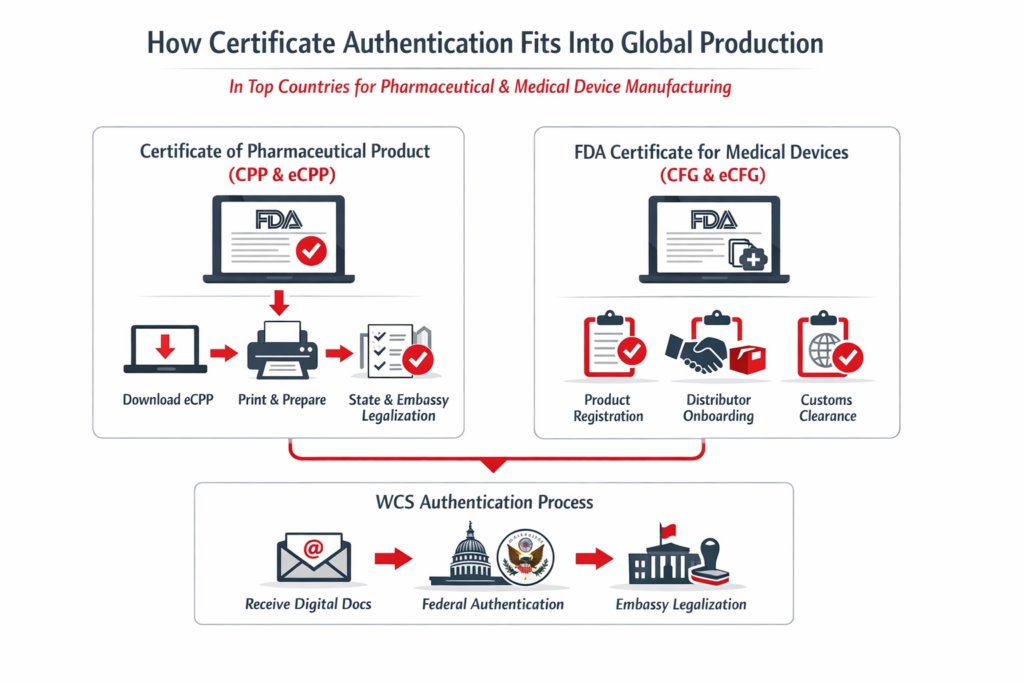
Certificates of Pharmaceutical Product (CPP) and Certificate to Foreign Government (CFG)
The FDA’s move to issuing electronic export certificates -electronic CPP (eCPP)and electronic CFG(eCFG)changed the mechanics, but not the need for authentication.
What that typically looks like operationally:
- Your team downloads the eCPP/eCFG from the FDA portal.
- The document is transmitted to your legalization partner in digital form.
- It is printed and prepared for federal authentication steps, then routed through any required embassy legalization.
How WCS supports these certificate workflows
Washington Consular Services (WCS) streamlines the entire export certificate process, from application through legalization or apostille, ensuring minimal turnaround time for our clients.
We can apply for an Export Certificate in one of two ways: as an authorized agent on behalf of the company, or directly through the company’s FDA account. This flexibility allows us to adapt to each client’s preferred setup and internal processes.
Once we receive a request for an export certificate, we process and submit the application to the FDA on the same day. Upon receiving the digital copy from the FDA, we immediately proceed with legalization and apostille — again, on the same day. This same-day, end-to-end approach eliminates unnecessary delays and ensures that product registrations and international filings stay on track.
If your team needs help authenticating business, regulatory, or trade documents for international use, Washington Consular Services (WCS) can support apostille and embassy legalization workflows to help keep cross-border timelines on track.
Key takeaways
- The top countries for pharmaceutical and medical device manufacturing combine scale, specialization, and regulatory readiness.
- Pharma and device manufacturing are concentrating in a mix of large markets and specialized hubs.
- The United States and Germany remain anchors for high-value, regulated production.
- China and India continue to scale, especially for volume and supply-chain depth.
- Ireland and Singapore draw investment for export-oriented and advanced manufacturing environments.
- Mexico and Costa Rica stand out in devices for nearshoring and established medtech clusters.
- Cross-border timelines often hinge on document readiness, not factory readiness.
Source links
- Grand View Research: https://www.grandviewresearch.com/horizon/outlook/biologics-manufacturing-market/united-states
- Germany Trade and Invest: https://www.gtai.de/en/invest/industries/healthcare-market-germany/pharmaceutical-industry
- Canton of Basel-Stadt: https://www.bs.ch/en/schwerpunkte/portrait/economie-et-travail/important-industries-and-companies
- IDA Ireland: https://www.idaireland.com/latest-news/insights/pharmaceutical-manufacturing-companies-ireland
- MedXDRG: https://medxdrg.com/understanding-where-the-pharmaceutical-industry-is-located-a-global-overview
- Singapore EDB: https://www.edb.gov.sg/en/our-industries/biotechnology-pharmaceuticals.html
- Electronic Manufacturing Service: https://electronicmanufacturingservice.org/where-are-most-medical-devices-manufactured/
- Alpha Sophia: https://www.alphasophia.com/blog-post/leading-medical-device-companies-by-country
- Tijuana EDC: https://tijuanaedc.org/tijuanas-medical-device-cluster-a-success-story/
- USITC executive briefing (PDF): https://www.usitc.gov/publications/332/executive_briefings/ebot_costa_rica_medical_devices.pdf
- International Trade Administration: https://www.trade.gov/country-commercial-guides/japan-medical-devices
- Medical Korea / KHIDI: https://www.medicalkorea.or.kr/en/aboutkhidi

